Abstract
Comprehensive proteomic studies of HSC derived from bone marrow of healthy human subjects (n = 59) in different age groups (range: 20 - 72 years) showed that aging HSCs are characterized not only by myeloid lineage skewing, senescence associated secretory phenotype (SASP), accumulation of reactive oxygen species (ROS), anti-apoptosis, but prominently by elevated glycolysis, glucose uptake, and accumulation of glycogen. This is caused by a subset of HSC that has become more glycolytic than others and not on a per cell basis. Subsequent comparative transcriptome studies of HSCs from human subjects >60 years versus those from <30 years have confirmed this association of elevated glycolysis with aging transcriptome signature.
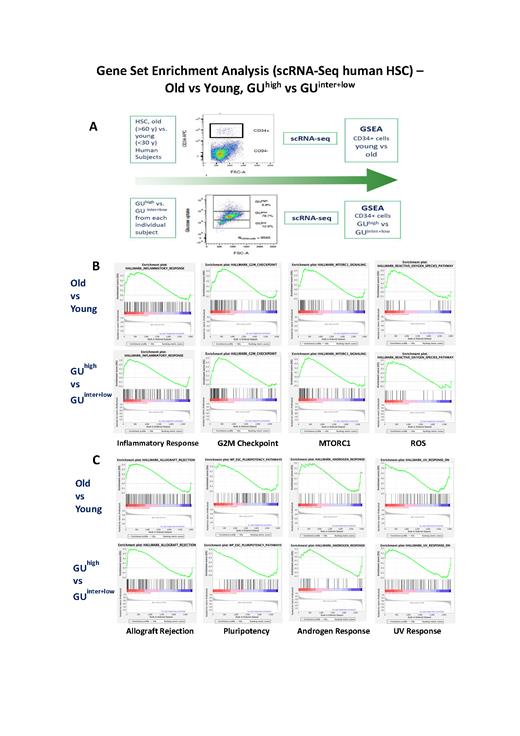
Provided with this background and based on glucose metabolism levels, we have developed a method to isolate human HSCs (CD34+ cells) from bone marrow into three distinct subsets with high, intermediate, and low glucose uptake (GU) capacity (GU high, GU inter, GU low). For human subjects >60 years old (n=9), the proportions of these subsets are: GU high= 5.4+3.5 %, GU inter= 66.4+22.5 %, GU low= 28.2+21.7 %. For subjects <30 years (n=5), the proportions are GU high= 1.7+1.5 %, GU inter= 66.5+36.9 %, GU low= 31.8+36.7. Single-cell RNA-sequencing (scRNA-seq) studies and gene ontology analysis of biological processes revealed that, compared to the GU inter and GU low subsets, the GU high cells showed a significantly higher expression of genes involved in myeloid development, inflammation response (AIF1, CASP2, ANXA1, ZFP36), anti-apoptosis (GSTP1, NME1, BCL2, DMNT1, BAX), cell cycle checkpoint (MCL1, CDK1, CDK4, EIF5A), histone regulation (BCL6, EGR1, KDM1A, MLLT3), b-galactosidase, and significantly lower expressions of genes involved in lymphoid development, and of MDM4, MDM2, FOXP1, SOX4, RB1. Functional studies indicated that the glycolytic enzymes were elevated in elderly HSCs, and the GU low subset corresponded to primitive and more pluripotent HSCs than the GU interand GU high subsets. Pathway analyses have then demonstrated that the GU high subset is associated with up-regulated p53 as well as JAK/STAT signaling pathways, characteristic of senescent HSCs observed in murine models. Applying Gene Set Enrichment Analysis (GSEA) algorithms, we have compared the scRNA-seq data of CD34+ cells derived from young (<30 years) versus older (>60 years) subjects, as well as the scRNA-seq data from GU high subset versus GU inter and GU lowsubsets from each individual subject (n = 6). The results are shown in Figure 1. In analogy to the comparison between old (>60 years) versus young (<30 years) HSCs (CD34+ cells), GSEA of the GU high versus GU inter and GU low subsets shows the same pattern of changes - significant upregulation of gene-set expressions for (a) inflammatory response (b) G2M checkpoint, (c) MTORC1, (d) ROS, (Fig. 1B), (e) allograft rejection; and down-regulation of gene-set expressions for (f) pluripotency, (g) androgen response, (h) UV response (Fig. 1C) as well as (i) interferon-a induction during SARS-CoV2-infection (data not shown in Fig. 1). Thus, our novel findings of elevated glycolysis coupled with significant activation of MTORC1 in the senescent cells of the HSC compartment have provided evidence for the important role of calorie restriction (CR) for healthy aging of HSCs. In numerous animal models, aging has been shown to be driven by the nutrient-sensing MTORC1 network. In animal models of aging, CR has been reported to deactivate the MTOR pathway, thus slowing aging and delaying diseases of aging.
Conclusion: In a series of multi-omics studies, we have demonstrated that the GU high subset is identical to the senescent cells (SCs) in human HSC compartment. Studies in animal models have shown that SCs in murine bone marrow are responsible for driving the aging process, and elimination of this subset by inhibitors of anti-apoptotic factors is able to rejuvenate hematopoiesis in mice. Our present results have provided cellular and molecular evidence that SCs in human HSC compartment are also dependent on anti-apoptotic factors, elevated MTORC1 as well as increased glycolysis for survival. Inhibition of MTORC1 or glycolysis, either by specific inhibitors or by CR, may eliminate senescent HSCs and promote rejuvenation of human hematopoiesis.
No relevant conflicts of interest to declare.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal